Along with life-giving photons, our sun emits energetic particles, electrons and protons. The earth’s magnetic field does a good job at protecting us (and all life) from the continuous bombardment of these particles. However, some still make it through, and our atmosphere is the next line of defense. The electrons and protons scatter, ionize, react with atoms of the upper atmosphere expending their harmful energy well above the surface of the earth. One method of interaction is the spallation (fancy way of saying blowing to smithereens) nitrogen and oxygen, leaving behind smaller nuclei. One particular nuclide is Be-7, and it happens to be radioactive. It has a half-life of about 53 days, so it sticks around long enough to work its way down from the upper atmosphere. Here are photos of a very simple experiment that needs only a surplus air sampler, a low-background gamma ray spectroscopy system, and nearly infinite patience.
This is a Staplex high-volume air sampler, with a 4-inch diameter filter paper mounted in it. I ran it outside for 2 hours a few weeks back. I purchase this air sampler at Surplus City in Albuquerque for $50, new in the box
Humorously, Staplex makes electric staplers & high speed air samplers. I really don’t see the synergistic connection.
It started out drawing a little more than 20 CFM of air through the paper.

After two hours, I removed the filter paper, folded it up and placed it on my HPGe for days, and days…
I counted it for five days, waiting until the radon daughters die away. Then I launched into an extremely long count on the filter paper, and an even longer background count.
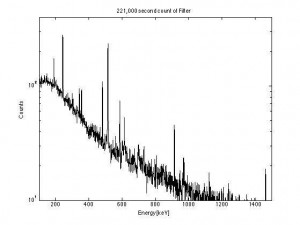
This is the result of a 221,000 second count (2.55 days) on the filter paper.
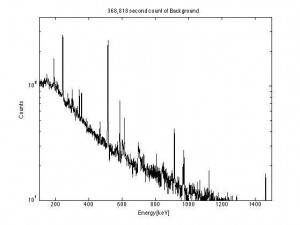
This is the result of a 368,818 second (4.25 day) background count. Note all the peaks despite being a “low background” – these are all from the building materials.
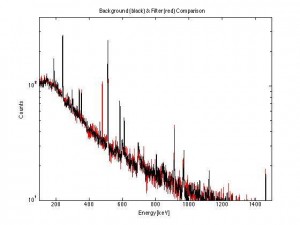
Performing a scaled comparison (filter paper in red, background in black) a single lone-red peak appears.
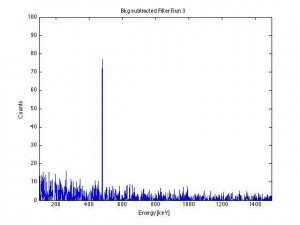
Subtract the background spectrum from the filter paper spectrum and a single 477 keV gamma line remains: Be-7.
The Be-7 concentration varies with the seasons, as thermals lift and descend the isotope. Note that there were only about 400 counts captured under this peak in 2.5 days.